Ritesh Panchoe and Rosanne Hertzberger, REBLAB / Group of Prof. Dr. Remco Kort, Systems Biology Lab, AIMMS, Vrije Universiteit Amsterdam, O|2 location De Boelelaan 1085, NL-1081HV Amsterdam, The Netherlands
Find the corresponding data file here…
Summary of the finding
After a pregrowth step on glucose, Lactobacillus crispatus strain RL_010 shows a significant lag phase on glycogen compared to glucose. This is consistent with the differential regulation of the glycogen degradation machinery which we reported before. Gardnerella vaginalis does not show such a difference in lag phase but does exhibit a long pause before starting growth on either glucose or glycogen.
Introduction
Previously we focused on growth of various strains of Lactobacillus crispatus on glycogen and glucose. We identified the role of a type 1 pullulanase, and we observed how glycogen breakdown ability is downregulated in the presence of glucose. Since a few months we have our anaerobic chamber up and running and we are now able to include Gardnerella vaginalis in our growth studies. Gardnerella belongs to the family Bifidobacteriaceae and is one of the prominent members of the dysbiotic vaginal community.
By studying glycogen metabolism in members of both the Lactobacillus-dominated and dysbiotic community we try to understand the fluctuations in vaginal glycogen content in reproductive-age women from a microbial perspective. In general, it is hypothesized that Lactobacillus utilizes glycogen as its primary carbon source to acidify the vagina. However, in the absence of a dominant Lactobacillus in the vaginal community (‘dysbiotic state’ or community state type IV) vaginal glycogen is reduced. We hypothesize that members of the dysbiotic vaginal community may be responsible for this enhanced glycogen degradation. The strong increase in absolute numbers of bacteria in the dysbiotic state increases carbon and energy requirements of the vaginal microbial community. During Ritesh’ internship we analyzed growth of Gardnerella vaginalis and Lactobacillus crispatus on glycogen to growth on glucose.
Methods
Gardnerella vaginalis DSM 4944 and Lactobacillus crispatus RL_10 were pregrown in liquid NYCIII medium [PDF] with 0,5% glucose and incubated anaerobically for 48 hours in an anaerobic chamber filled with a gas mix of 95% N2 and 5% CO2. Find full inoculation details here [PDF], in these experiments we inoculated anaerobically.
This preculture was tenfold diluted in NYCIII medium containing either 0,5% glucose, 0,5% glycogen or water as a control in flat-bottom transparent 96 well plate (Greiner 655161). The volume in each well was 380 uL to leave as little headspace as possible. The plates were sealed-off with adhesive film (VWR 60941-072).
This experiment should not be considered as anaerobic. Although the 96-well plate was prepared in the anaerobic chamber, it was subsequently taken to the plate reader outside of the chamber. It is safe to assume that part of this growth experiment should be considered aerobic.
We performed this experiment on three separate occasions and show the results of two individual experiments in the graph.
Results
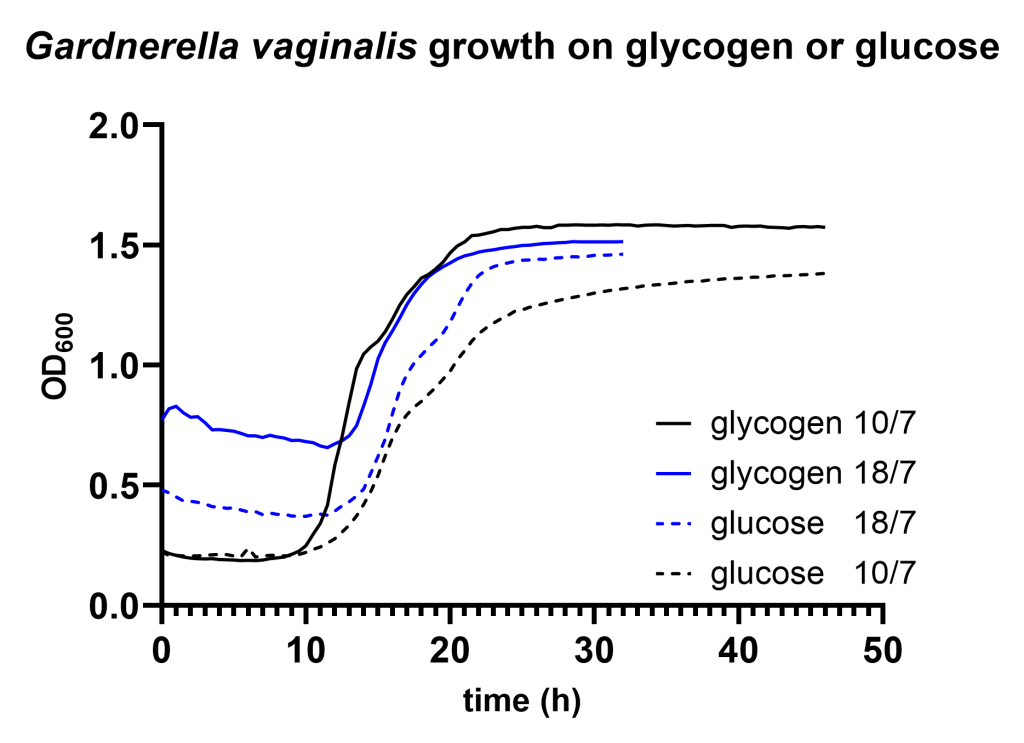
We find that Lactobacillus crispatus shows a considerable longer lag phase on glycogen compared to glucose after it has been pregrown on glucose (Figure 1A). Although growth of Gardnerella vaginalis resulted in a very long lag phase (>10 hours) on both carbon sources, we did not detect any difference in lag phase between growth on glycogen compared to glucose (Figure 1B).
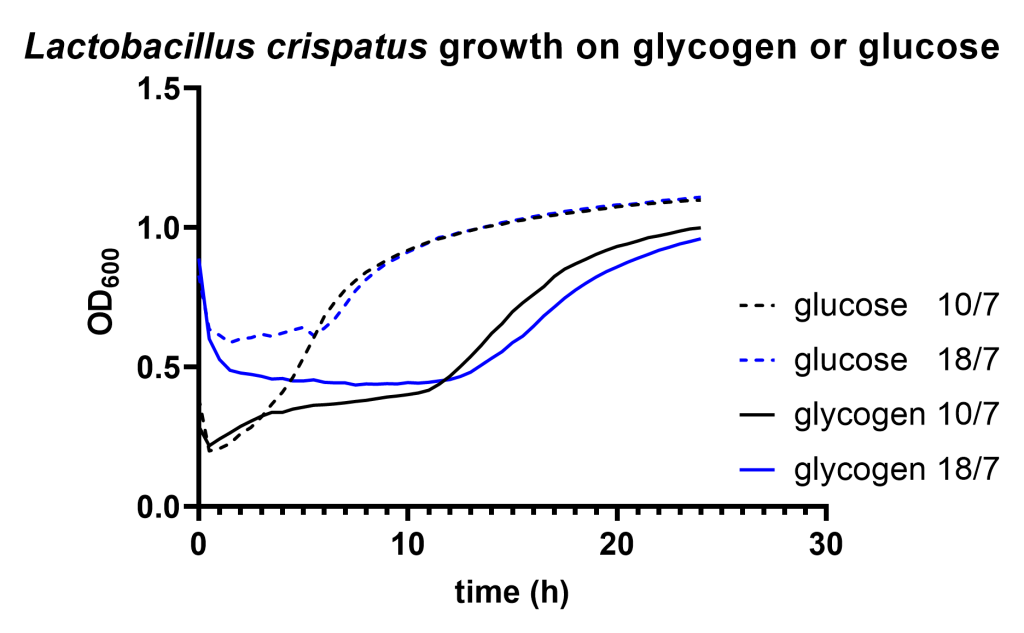
The lag phase of Lactobacillus crispatus may be explained by the repressed expression of the type 1 pullulanase in the presence of glucose. There are several other findings that support carbon catabolite regulation of this gene associated with glycogen metabolism: 1) previously, we were unable to detect glycogen degradation activity in L. crispatus pellets after growth on glucose. 2) The type 1 pullulanase gene of L. crispatus has a palindromic region upstream of its start site showing similarities to carbon responsive elements in Lactobacillus plantarum https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5986886/. 3) The expression of the ortholog pullulanase gene in L. acidophilus is also repressed by glucose. None of these findings provide conclusive evidence but they are all in line with repressed expression of the type 1 pullulanase in presence of glucose.
The ~10 hours lag phase of Gardnerella vaginalis on glycogen is likely due to other circumstances, for instance oxidative stress due to influx of oxygen through the microplate seal. The results do not exclude that Gardnerella regulates its genes involved in glycogen degradation, but under these experimental conditions Gardnerella grows as readily on glycogen as on glucose.
-Warning, wild speculation ahead- This difference in regulation of the glycogen degradation machinery of these bacterial species, that are associated with distinct microbial signatures of the vagina, may have implications on the bacterial dynamics in the human vagina. If a L. crispatus culture encounters glucose in the vagina, it is likely that it will continue growth on glucose and thereby stop expressing its enzymes needed for glycogen breakdown.
After depletion of glucose the bacteria will have to switch back to using vaginal glycogen but need some time to accomplish this metabolic switch. In contrast: if we find in further experiments that Gardnerella vaginalis constitutively expresses its glycogen degradation enzymes, these bacteria can continue growing on glycogen and may even be capable of utilizing both glycogen and glucose in paralel. It may even be beneficial for Gardnerella in bacterial competition to leave trace amounts of glucose during glycogen degradation (“chew more than you eat”), to provoke this metabolic shift in Lactobacillus crispatus.
Next experiments
-Alexander Woudstra is currently repeating these experiments in the anaerobic chamber. Although we do not have a plate reader in there, we do have a 37° heating block.
-A simple experiment that would confirm the role of glycogen in these growth dynamics is by pre-growing on glycogen. This should eliminate the L crispatus lag-phase on glycogen.
-Lastly, Alexander is also attempting to grow these bacteria on both glucose and glycogen. If our hypothesis is correct, Lactobacillus crispatus should degrade glucose first and then switch to glycogen. Gardnerella vaginalis should be capable of utilizing both substrates simultaneously.
Practical notes on the experiments:
The data file has several other experimental conditions that were included. For instance: growth of Lactobacillus iners, growth on galactose and maltose and growth of Gardnerella vaginalis supplemented with supernatants from previous growth experiments. None of these experimental conditions provided results that we would like to discuss extensively at this point.
Ritesh has repeated this experiment on one more day (July 16th), but during this experiment the controls were contaminated. Also, for some unknown reason L crispatus did not show growth on glycogen during this experiment. We have omitted these lines from the graph but the data can be found in a separate tab in the data file.
The increased OD600 measurements at the onset of some growth experiments were likely due to air bubbles under the plate seal.
Some more practical findings from his internship:
-the anaerobic chamber is key in growing Gardnerella vaginalis and Lactobacillus iners. Previously we were attempting to grow them in anaerobic jars, but this means that the handling and pipetting is happening in the presence of oxygen and this means rapid decline of viability. In this project we used the anaerobic chamber for the first time and it worked out well.
-However, we are still experiencing problems with reproducibility of growth of Lactobacillus iners. We still have to find the best way to capture the exponential phase and the optimal inoculation conditions.
-Starch degradation was in this project much more heterogeneous as previously. We need to look into the origins of this heterogeneity, and we think it may have something to do with handling of the cultures. I believe that the findings from Deborah’s internship about starch degradation are still credible and reproducible, but the difference between experiments in this project were much larger and we do not feel confident enough to share them.
-Due to these practical reasons we chose to only share this one observation on growth of Lactobacillus crispatus and Gardnerella vaginalis.
Thanks to
We would like to express our gratitude to Jurgen Haanstra for supervision, advise and positive vibes.